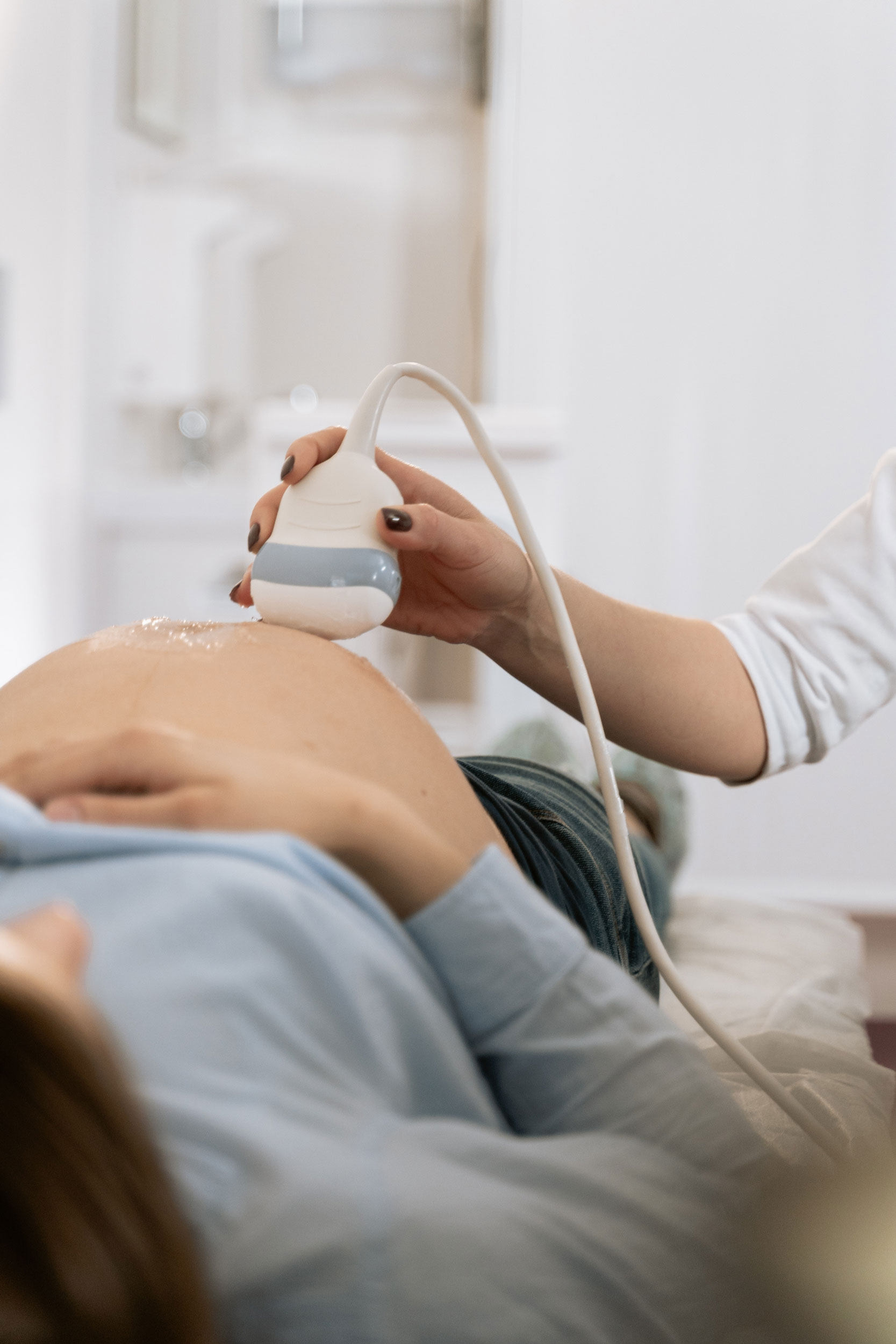
The Elle TENS range is Australia's most trusted, reliable and recognised electrotherapy devices, for labour, incontinence, general pain relief and injury recovery.
Shop by Collections
For Labour
TENS machines for drug free pain relief before, during and after birth.
For Incontinence
Pelvic floor stimulators, helping men and women to take back control.
For Pain Relief
Relieve knee, neck and back pain and with our TENS machines.
All Products
All TENS devices, and accessories for all TENS machines.
Shop Our Products
Happy Customers
Making a difference to your health and wellbeing. Health Rebates for TENS Machines in Australia





















Learn About Elle TENS Devices

How does a TENS machine work?
TENS is Transcutaneous Electrical Nerve stimulation or TENS for short. Find out more.TENS for Pregnancy & Labour
Planning for giving birth is a physical, mental and emotional journey. Find out more.TENS/EMS for Pain Relief
Electrical Muscle Stimulation (EMS) is a technique to stimulate muscles with an electric current to achieve a specific result.Pelvic Floor Stimulators for Incontinence
Incontinence – what is this big bad scary word? Find out more about how our Pelvic Floor Stimulators can help.Frequently Asked Questions
Yes! Our award winning Elle TENS is a great addition to your birthing kit. It can be used from early labour at home, or all the way through until birth. It also allows you to stay home from the hospital for longer or support you during your home birth.
In certain circumstances a TENS machine can be used during pregnancy for pain relief, but it is important to consult with a healthcare provider first and only under their supervision as they are familiar with your pregnancy.
A TENS (Transcutaneous Electrical Nerve Stimulation) machine works by delivering low-frequency electrical pulses to the nerves through electrodes placed on the skin. This pulse overloads your nervous system. As your body can only focus on so much information at a time, it prioritises the TENS pulse (nerve activity) over pain signals, meaning that fewer pain signals make it to your brain and spinal cord, so you feel less pain. This is a part of ‘The Gate Control Theory of Pain’.
At the same time the body will release natural pain relieving endorphins.
As soon as contractions start, it’s time to pop your Elle TENS on so you can start to build endorphins. Our built-in contraction timer can help you also to keep track of your contractions as your labour progresses.
It is best to test your tens before your labour begins, so that you know where to correctly place the electrode pads and familiarise yourself with how it works. Once your electrodes are in the correct place, turn the machine on, starting with the lowest settings and gradually turn up each side. Remember the boost button is your best friend during labour as contractions get stronger you may not need to use it initially.
Of course! Your Elle TENS can be used on its own all the way until you give birth, or with other forms of pain relief as needed.
There are no known side effects associated with using a TENS machine during labour, it is a safe drug free pain relief option for you and your baby recommended by medical professionals including midwives, OBs, physios etc
The Elle TENS should not be used in the shower or bath. When mixing up your labour techniques and having a shower for example, remember to pop the electrodes/pads back on the paper they came on so they are ready to use again. It can be helpful to cycle through your labour tools but our mums tell us they want to pop the Elle TENS back on again straight after a shower or bath.
We would love you to have your TENS ready to go by 34-36 weeks. This gives you peace of mind that you have your labour tool kit ready to go for when your baby decides it’s time!
Yes, the Elle TENS is certainly one of the best labour tools you can use during your home birth. Check out our reviews on how Mums have used the Elle TENS through their entire labour at home.
We definitely recommend this! You can test your TENS by placing the electrodes on your arm and turning it to a low setting. You can also ask your birth partner to learn where to put the electrodes on your back, check out electrode placement here.
This will help you get a sense of how the machine feels or troubleshoot any issues that may arise. PS make sure to pop the electrodes back on the paper they came on when not in use so they are in top condition for when you need them again.
Of course, sometimes baby can be in a position which is not the most comfortable for you. The Elle TENS is there to help you and will provide that much needed pain relief.
Yes, your Elle TENS machine can be used during all stages of labour, from early labour right through to birth. Remember that Elle TENS Opti-max key we mentioned before? This will allow you to increase intensity of the pulses as you progress through the different stages of labour putting you in control of your pain.
Most definitely. Your Elle TENS comes with a neck strap to enable you to move around and change positions as you need.
Subscribe to our newsletter and get the latest trending products and offers updates.